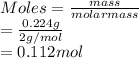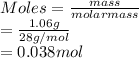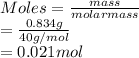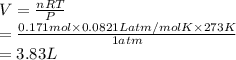Chemistry, 03.06.2021 01:00 sylviaswanson1
A mixture of 0.224 g of H2, 1.06 g of N2, and 0.834 g of Ar is stored in a closed container at STP. Find the volume (in L) of the container, assuming that the gases exhibit ideal behavior.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 19:30
Determine the number o moles of ions/atoms/particle in the following: 2.50 miles of k2s (let me know how to do)
Answers: 1

Chemistry, 21.06.2019 20:00
Drag each number to the correct location on the equation. each number can be used more than once, but not all numbers will be used. balance the equation with the coefficients. 2 3 4 5 kclo3 -> kcl + o2
Answers: 1

Chemistry, 22.06.2019 08:30
Sally is making a model of a magnesium atom with an atomic mass number of 24 for her chemistry class. she has foam balls for the protons, neutrons, and electrons. she has added 6 neutrons to her model so far. how many more neutrons does she need to add to complete her neutral atom of magnesium?
Answers: 1

Chemistry, 22.06.2019 18:30
How many moles of bromine are needed to produce 3.23 moles of potassium bromide
Answers: 1
You know the right answer?
A mixture of 0.224 g of H2, 1.06 g of N2, and 0.834 g of Ar is stored in a closed container at STP....
Questions


Mathematics, 28.11.2019 22:31









Computers and Technology, 28.11.2019 22:31

Mathematics, 28.11.2019 22:31








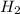 = 0.224 g
= 0.224 g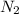 = 1.06 g
= 1.06 g