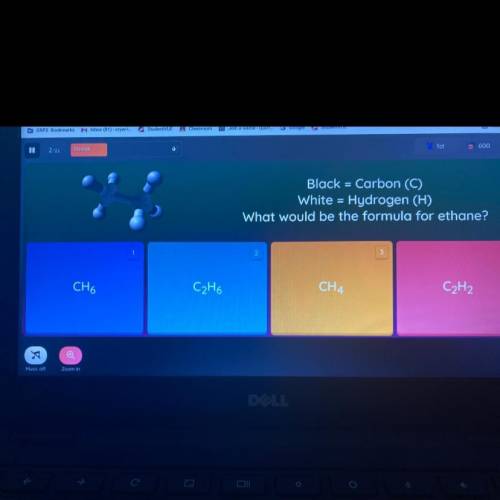
What would be the formula for ethane?
...

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 17:10
Nitric oxide (no) can be formed from nitrogen, hydrogen and oxygen in two steps. in the first step, nitrogen and hydrogen react to form ammonia: n2(g) + 2 h_2(g) rightarrow 2 nh_3 (g) delta h = -92. kj in the second step, ammonia and oxygen react to form nitric oxide and water: 4 nh_3(g) + 5 o_2(g) rightarrow 4no(g) + 6 h_2o(g) delta h = -905. kj calculate the net change in enthalpy for the formation of one mole of nitric oxide from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 1

Chemistry, 22.06.2019 06:00
Ethanol (c2h5oh) is produced from the fermentation of sucrose in the presence of enzymes. c12h22o11(aq) + h2o(g) 4 c2h5oh(l) + 4 co2(g) determine the theoretical yield and the percent yields of ethanol if 680. g sucrose undergoes fermentation and 326.5 g ethanol is obtained. theoretical _ g _ percent %
Answers: 1

Chemistry, 22.06.2019 06:30
If 1.8 l of water is added to 2.5l of a 7.0 m koh solution, what is the molarity of the new solution
Answers: 1

Chemistry, 22.06.2019 09:00
Astudent is asked to identify and element that is pale yellow brittle solid and does not conduct electricity. at which location in this periodic table would the element most likely be found?
Answers: 2
You know the right answer?
Questions

Mathematics, 21.10.2019 17:30

Mathematics, 21.10.2019 17:30


Mathematics, 21.10.2019 17:30

Mathematics, 21.10.2019 17:30

Mathematics, 21.10.2019 17:30


Mathematics, 21.10.2019 17:30

Biology, 21.10.2019 17:30

Biology, 21.10.2019 17:30

Mathematics, 21.10.2019 17:30



World Languages, 21.10.2019 17:30


Mathematics, 21.10.2019 17:30

Mathematics, 21.10.2019 17:30

Mathematics, 21.10.2019 17:30

Biology, 21.10.2019 17:30

Mathematics, 21.10.2019 17:30