Chemistry, 05.06.2021 14:00 winterblanco
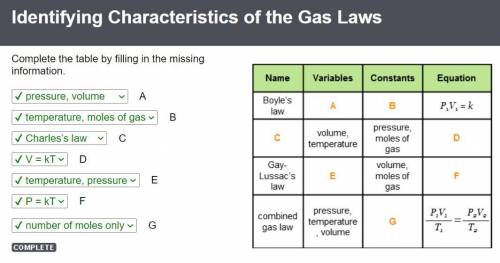
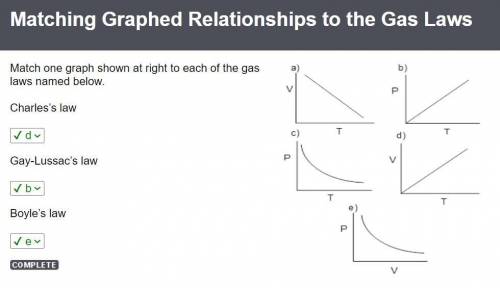
If you decreased the volume of a sample of gas by a factor of three while maintaining a constant pressure, how would the absolute temperature of the gas be affected?
A gas at 300 K and 4.0 atm is moved to a new location with a temperature of 250 K. The volume changes from 5.5 L to 2.0 L. What is the pressure of the gas at the new location?
What is the partial pressure of 0.50 mol Ne gas combined with 1.20 mol Kr gas at a final pressure of 730 torr?

Answers: 1
Another question on Chemistry


Chemistry, 21.06.2019 19:00
Which term best describes the form sound takes as it travels away from a drum (a- gas)(b-music) ( c-waves) (d-particles
Answers: 3

Chemistry, 22.06.2019 07:20
The diagrams show objects’ gravitational pull toward each other. which statement describes the relationship between diagram x and y? gravity attracts only larger objects toward one another. gravity attracts larger objects only if they are close to one another. if the masses of the objects increase, then the force between them also increases. if distance between the objects increases, then the amount of force also increases.
Answers: 1

Chemistry, 22.06.2019 23:30
If it is an isoelectronic series select true, if not select false. o2-, s2-, se2-, te2- na+, k+, rb+, cs+ n3-, p3-, as3-, sb3- ag, cd+, sn3+, sb4+ f-, cl-, br-, i- f-, ne, na+, mg2+ s2-, s, s6+
Answers: 1
You know the right answer?
If you decreased the volume of a sample of gas by a factor of three while maintaining a constant pre...
Questions



Chemistry, 12.04.2021 14:00


Chemistry, 12.04.2021 14:00

English, 12.04.2021 14:00


Computers and Technology, 12.04.2021 14:00

English, 12.04.2021 14:00

English, 12.04.2021 14:00

Social Studies, 12.04.2021 14:00

English, 12.04.2021 14:00


Spanish, 12.04.2021 14:00


Mathematics, 12.04.2021 14:00

English, 12.04.2021 14:00


Computers and Technology, 12.04.2021 14:00

Mathematics, 12.04.2021 14:00