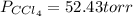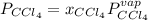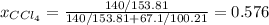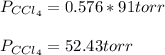At a certain temperature the vapor pressure of pure chloroform (CHCl3) is measured to be 91. torr. Suppose a solution is prepared by mixing 140. g of chloroform and 67.1 g of heptane (C, H16) of chloroform and 67.1 g of heptane (C7H16 Calculate the partial pressure of chloroform vapor above this solution.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 20:30
There is an area in idaho named craters of the moon where most of the ground is covered with basalt, adark gray, igneous rock with no visibl crystals. what can you infer about the geographical history of the area?
Answers: 1

Chemistry, 22.06.2019 14:30
In water, a strong acid will break down into its component parts. a. completely b. partly c. never in water, a weak base will break down into its component parts. a. completely b. partly c. never
Answers: 2

Chemistry, 22.06.2019 18:50
Asample of tin (ii) chloride has a mass of 0.49 g. after heating, it has a mass of 0.41 g. what is the percent by mass of water in the hydrate? %
Answers: 1

Chemistry, 23.06.2019 01:20
Use the de broglie's wave equation to find the wavelength of an electron moving at 4.5 × 106 m/s. show your work. note: h= plank's constant (6.62607 x 10-34 j s)
Answers: 1
You know the right answer?
At a certain temperature the vapor pressure of pure chloroform (CHCl3) is measured to be 91. torr. S...
Questions

Mathematics, 18.11.2020 17:50

English, 18.11.2020 17:50





Computers and Technology, 18.11.2020 17:50

History, 18.11.2020 17:50



Biology, 18.11.2020 17:50

Mathematics, 18.11.2020 17:50

Biology, 18.11.2020 17:50

Mathematics, 18.11.2020 17:50

Mathematics, 18.11.2020 17:50




Spanish, 18.11.2020 17:50