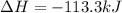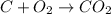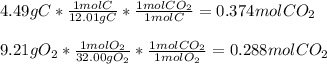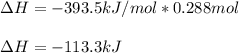Chemistry, 13.06.2021 04:20 linshweyioo9931
The standard enthalpy of formation of CO2(g) is -393.5 kJ mol-1 . What is the enthalpy change if 4.49 g C(s) reacts with 9.21 O2(g) to form CO2(g)

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 01:50
Ase your answer to this question on the information below.hydrocarbons and fissionable nuclei are among the sources used for the production of energy in the united states. a chemical reaction produces much less energy than a nuclear reaction per mole of reactant.the balanced chemical equation below represents the reaction of one molecule of a hydrocarbon with two molecules of oxygen.chemical equation: ch4 + 2o2 → co2 + 2h2o + 1.48 × 10−18 jthe nuclear equation below represents one of the many possible reactions for one fissionable nucleus. in this equation, x represents a missing product.nuclear equation: write an isotopic notation for the missing product represented by x in the nuclear equation.
Answers: 1

Chemistry, 22.06.2019 08:00
Me i dont know what to do! the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1

Chemistry, 22.06.2019 08:30
Sally is making a model of a magnesium atom with an atomic mass number of 24 for her chemistry class. she has foam balls for the protons, neutrons, and electrons. she has added 6 neutrons to her model so far. how many more neutrons does she need to add to complete her neutral atom of magnesium?
Answers: 1

You know the right answer?
The standard enthalpy of formation of CO2(g) is -393.5 kJ mol-1 . What is the enthalpy change if 4.4...
Questions




Mathematics, 22.11.2021 19:10

Biology, 22.11.2021 19:10

Mathematics, 22.11.2021 19:10





Mathematics, 22.11.2021 19:10




Social Studies, 22.11.2021 19:10


Mathematics, 22.11.2021 19:10

Advanced Placement (AP), 22.11.2021 19:10

Mathematics, 22.11.2021 19:10

Mathematics, 22.11.2021 19:10