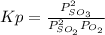Chemistry, 14.06.2021 19:40 hihudgins902
Consider the following reaction at 298 K.
2 SO2(g) + O2(g) → 2 SO3(g)
An equilibrium mixture contains O2(g) and SO3(g) at partial pressures of 0.43 atm and 2.6 atm, respectively. Using data from Appendix 4, determine the equilibrium partial pressure of SO2 in the mixture.
atm.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 09:20
Sugar is dissolved in water. which is the solute? sugar neither both water
Answers: 1

Chemistry, 22.06.2019 11:20
Which of the following contributes to the structural rigidity of cellulose? adjacent glucose polymers are stabilized by hydrogen bonding. glucose residues are joined by (α1→4) linkages. cellulose is a highly branched molecule. the conformation of the glucose polymer is a coiled structure.
Answers: 2

Chemistry, 22.06.2019 13:10
Select the correct answer a modure consists of glucose and water. what is the percent composition of glucose in the mixture if it contains 1.3 moles of glucose (cho total mass of the mature is 276 grams? ) and the a 1775
Answers: 1

Chemistry, 22.06.2019 20:10
What would happen to a volleyball left outside in the winter? o o o o a. it would expand. b. it would lose air. c. it would shrink. d. it would explode.
Answers: 2
You know the right answer?
Consider the following reaction at 298 K.
2 SO2(g) + O2(g) → 2 SO3(g)
An equilibrium mixture...
An equilibrium mixture...
Questions





English, 21.11.2020 22:40

Health, 21.11.2020 22:40

Mathematics, 21.11.2020 22:40

Mathematics, 21.11.2020 22:40



Mathematics, 21.11.2020 22:40

Mathematics, 21.11.2020 22:40

Mathematics, 21.11.2020 22:40

History, 21.11.2020 22:40

Mathematics, 21.11.2020 22:40

Physics, 21.11.2020 22:40



Mathematics, 21.11.2020 22:40