
Chemistry, 14.06.2021 23:30 lindseydupre
Pressurized metal gas cylinders are generally used to store commonly used gases in the laboratory. At times, it can be easier to chemically prepare occasionally used gases. For example, oxygen gas can be prepared by heating KMnO4(s) according to the following chemical reaction:
2KMnO4(s) → K2MnO4(s) + MnO2(s) + O2(g)
How many grams of KMnO4 would you need to produce 0.27 moles of O2, assuming 100% conversion?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 10:30
Which characteristics can be used to differentiate star systems? check all that apply.
Answers: 2

Chemistry, 22.06.2019 12:00
What does a complete balanced chemical equation include? a. exothermic coefficients b. endothermic coefficients c. valence electrons d. molar coefficients
Answers: 1

Chemistry, 22.06.2019 14:30
Calculate the mass of carbon in 97.0 g of sucrose c12h22o11
Answers: 3

Chemistry, 23.06.2019 03:00
Which of the following is a chemical property of water at 4 c
Answers: 2
You know the right answer?
Pressurized metal gas cylinders are generally used to store commonly used gases in the laboratory. A...
Questions



Chemistry, 29.08.2020 23:01

Social Studies, 29.08.2020 23:01


History, 29.08.2020 23:01


Business, 29.08.2020 23:01












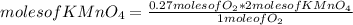
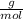 .
. 

