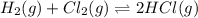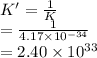The equilibrium constant (K) for the reaction
2HCI(g) = H2(g) + Cl2(g)
is 4.17 x 10-34 at 25°...


Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 19:10
When will le chatelier's principle come into effect? at the beginning of a reaction, when there are only reactants when a reaction has reached chemical equilibrium when a catalyst is added to a reaction mixture when a reaction is occurring but not yet at equilibrium
Answers: 3

Chemistry, 22.06.2019 02:00
The alkali metals (group 1) consist of lithium (3), sodium (11), potassium (19), rubidium (37), cesium (55), and francium (87). they are soft, metallic solids with low densities and low melting points. based on the data shown in figure 1, how many valence electrons do alkali metals share?
Answers: 3

Chemistry, 22.06.2019 06:30
Summarize possible ways in which phases of matter could combine to form a solution.
Answers: 2

Chemistry, 22.06.2019 09:30
1. explain hydrogen peroxide, h 2 o 2 properties and decomposition reaction. 2. describe how each of the following natural cycles plays a part in earth’s climate system. (a) the water cycle (b) the carbon cycle
Answers: 1
You know the right answer?
Questions

History, 26.05.2020 18:01



Mathematics, 26.05.2020 18:01


History, 26.05.2020 18:01


Social Studies, 26.05.2020 18:01

Chemistry, 26.05.2020 18:01



Physics, 26.05.2020 18:01




Mathematics, 26.05.2020 18:01


Social Studies, 26.05.2020 18:01

History, 26.05.2020 18:01

 .
. ... (1)
... (1)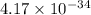 .
.