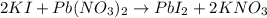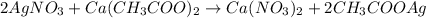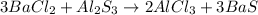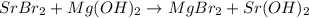Which reaction produces an insoluble product?
A
2KI + Pb(NO )2 → → PbI, + 2KNO
B
...

Chemistry, 15.06.2021 09:10 christylivingsowzxa2
Which reaction produces an insoluble product?
A
2KI + Pb(NO )2 → → PbI, + 2KNO
B
2AgNO3 + Ca(CH,02)2 + Ca(NO3)2 + 2AgC, H,O,
C
3BaCl2 + ALLS
→ 2AlCl3 + 3BaS
.
D
SrBr, + Mg(OH)2 → MgBr, + Sr(OH)2

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 03:50
Consider the reaction: n2(g) + o2(g) ? 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 09:30
1. explain hydrogen peroxide, h 2 o 2 properties and decomposition reaction. 2. describe how each of the following natural cycles plays a part in earth’s climate system. (a) the water cycle (b) the carbon cycle
Answers: 1

Chemistry, 22.06.2019 19:00
Avolleyball player hit a ball with a mass of 0.25 kg. the average acceleration of the ball is 15.5 m/s². how much force did the volleyball player apply to the ball? 62.0 n 3.87 n 62.0 m/s² 3.87 m/s²
Answers: 2
You know the right answer?
Questions

Arts, 17.02.2021 21:10


Mathematics, 17.02.2021 21:10


Advanced Placement (AP), 17.02.2021 21:10


Chemistry, 17.02.2021 21:10

Mathematics, 17.02.2021 21:10

Mathematics, 17.02.2021 21:10


Mathematics, 17.02.2021 21:10



Mathematics, 17.02.2021 21:10

Mathematics, 17.02.2021 21:10


Physics, 17.02.2021 21:10


Biology, 17.02.2021 21:10

Mathematics, 17.02.2021 21:10