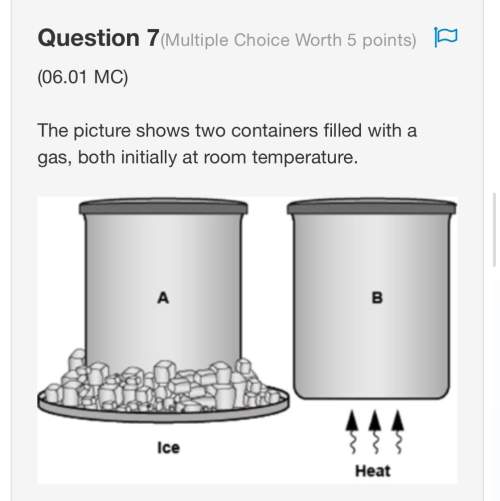
8.2 14,5 g of iron reacts with 12 g of water during a corrosion reaction to form
Fe3O4 and H2, according to the following equation.
Fe + H2O + Fe3O4 + H2
8.2.1 Balance the equation.
8.2.2 Find the substance which is the limiting reactant? Show your
calculations.
8.2.3 Using the calculations in your answer to Question 8.2.2, determine
the mass of Fe3O4 that will be formed.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 01:20
Match the acid base pairs by arranging the acid name with the conjugate base formula. hydrogen carbonate hydrogen phosphate carbonic acid read water sulfuric acid phosphoric acid a. co32- b. hso4- c. hco3- d. po43- e. h2po4- f. oh-
Answers: 1


Chemistry, 22.06.2019 12:00
Marcel just purchased 1.69 grams of iron fillings in order to make living putty for his 6 year old niece. how many moles of iron are made in his sample?
Answers: 1

Chemistry, 22.06.2019 19:00
What information does a complete ionic equation give that the balanced equation doesn’t show?
Answers: 1
You know the right answer?
8.2 14,5 g of iron reacts with 12 g of water during a corrosion reaction to form
Fe3O4 and H2, acco...
Questions





Chemistry, 07.04.2021 23:30

Mathematics, 07.04.2021 23:30


Computers and Technology, 07.04.2021 23:30

Chemistry, 07.04.2021 23:30



Mathematics, 07.04.2021 23:30

Chemistry, 07.04.2021 23:30


Social Studies, 07.04.2021 23:30

English, 07.04.2021 23:30

Mathematics, 07.04.2021 23:30



Biology, 07.04.2021 23:30