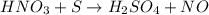Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 09:50
Although respiratory organs vary across different organisms, they all contain respiratory surfaces that have a large surface area and are extremely thin. explain why having an extremely thin respiratory surface with a large surface area is advantageous for the process of gas exchange
Answers: 1

Chemistry, 22.06.2019 13:00
16. why must the number of electrons lost equal the number of electrons gained in every redox reaction? use 3 – 4 sentences in your own words to address this question. 18. what type of radiation is emitted when chromium-51 decays into manganese-51? show the nuclear equation that leads you to this answer. 19. a radioactive nucleus alpha decays to yield a sodium-24 nucleus in 14.8 hours. what was the identity of the original nucleus? show the nuclear equation that leads you to this answer.
Answers: 2

Chemistry, 22.06.2019 18:00
Which three statements represent the benefits of performing experiments using computer simulations?
Answers: 3

Chemistry, 22.06.2019 21:30
What is the effect of returning nuclear reactor cooling water back into bodies of water?
Answers: 3
You know the right answer?
HNO3 + S --> H2SO4 + NO Now identify the element oxidized and the element reduced. Which element...
Questions

Mathematics, 05.05.2022 03:10


English, 05.05.2022 03:10


Mathematics, 05.05.2022 03:10


History, 05.05.2022 03:10


Business, 05.05.2022 03:10


Biology, 05.05.2022 03:10






Chemistry, 05.05.2022 03:10



Mathematics, 05.05.2022 03:10