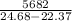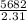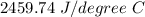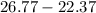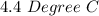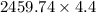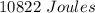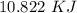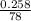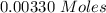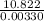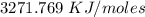Chemistry, 16.06.2021 23:10 ejcastilllo
Two experiments were conducted in a bomb calorimeter. The first one to determine the heat capacity of the calorimeter, the second the heat of combustion of the carcinogenic substance benzene (C6H6). a. In the first experiment, the temperature rises from 22.37 o C to 24.68 o C when the calorimeter absorbs 5682 J of heat. Determine the heat capacity of the calorimeter. Page 3 of 4 b. In the second experiment, the combustion of 0.258 g of benzene increases the temperature from 22.37 o C to 26.77 o C. Determine the heat of combustion for 1 mol of benzene.

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 03:00
Match term definition ellipse a) diagonal cross section of a cylinder circle b) diagonal cross section through the widest part of a sphere sphere c) cross section parallel to the base of a cone great circle d) shape created when a semi-circle is rotated around the y-axis triangle e) perpendicular cross section of a cone
Answers: 1

Chemistry, 22.06.2019 12:30
If anyone would be able to me out with these three questions it would be these are from the chem 2202 course.
Answers: 3

Chemistry, 22.06.2019 22:30
Amedication is given at a dosage of 3.000 mg of medication per kg of body weight. if 0.1500 g of medication is given, then what was the patient's weight in pounds (lbs)? there are 453.59g in 1 lb.
Answers: 2
You know the right answer?
Two experiments were conducted in a bomb calorimeter. The first one to determine the heat capacity o...
Questions

Mathematics, 10.02.2021 21:00




Mathematics, 10.02.2021 21:00



Mathematics, 10.02.2021 21:00



English, 10.02.2021 21:00


Mathematics, 10.02.2021 21:00

Biology, 10.02.2021 21:00

Mathematics, 10.02.2021 21:00


Mathematics, 10.02.2021 21:00



Social Studies, 10.02.2021 21:00