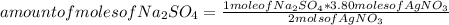Silver sulfate is precipitated from solution according to the following reaction 2 AgNO3 (aq) Na2SO4 (aq) --> Ag2SO4 (s) 2 NaNO3 (aq) If 3.80 moles of AgNO3 and 3.05 moles of Na2SO4 are present initially, how many moles of excess reactant will remain after precipitation is complete

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 18:10
The enthalpy of formation of water is -285.8 kj/mol. what can be inferred from this statement?
Answers: 1

Chemistry, 22.06.2019 15:30
The reactions of photosynthesis occur in the of plant cell? a.mitochondria. b. lysosomes. c. chloroplasts. d. chlorophyll
Answers: 1


Chemistry, 22.06.2019 18:30
Which of the following words describe the reality that the universe looks the same from various perspective
Answers: 3
You know the right answer?
Silver sulfate is precipitated from solution according to the following reaction 2 AgNO3 (aq) Na2SO4...
Questions


Mathematics, 01.03.2021 19:00

Mathematics, 01.03.2021 19:00

Mathematics, 01.03.2021 19:00


Computers and Technology, 01.03.2021 19:00



Mathematics, 01.03.2021 19:00



Mathematics, 01.03.2021 19:00

Biology, 01.03.2021 19:00

Mathematics, 01.03.2021 19:00

Physics, 01.03.2021 19:00

Chemistry, 01.03.2021 19:00


English, 01.03.2021 19:00