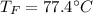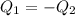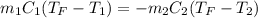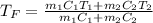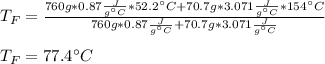Two objects are brought into contact Object 1 has mass 0.76 kg, specific heat capacity 0.87) g'c and initial temperature 52.2 'C. Object #2 has mass 70.7 9. specific heat capacity 3.071" "c' and initial temperature 154 *C. What is the final temperature of the two masses after thermal equilibrium has been reached? Assume the two objects are thermally isolated from everything else. Express your answer in C

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:00
Acurium-245 nucleus is hit with a neutron and changes as shown by the equation. complete the equation by filling in the missing parts. 52
Answers: 2

Chemistry, 22.06.2019 17:00
How can a give a full method for the experiment of separating sand from water by filtration? 1-materials 2-steps 3-conclusion also for water and salt separated by the evaporation or distillation process
Answers: 1


Chemistry, 22.06.2019 18:00
What amount of heat is exchanged when 106.2 grams of substance y goes from a liquid at 35 degrees celsius to a solid at the same temperature? melting point of substance y = 35 degrees c; δhvaporization = 3.67 j/mol; δhfusion = 3.30 j/mol. mwsubstance y = 28.22 g/mol. −12.4 j −3.51 x 102 j 1.24 x 101 j 351 j
Answers: 1
You know the right answer?
Two objects are brought into contact Object 1 has mass 0.76 kg, specific heat capacity 0.87) g'c and...
Questions

English, 29.10.2020 06:00

Geography, 29.10.2020 06:00



Mathematics, 29.10.2020 06:00



Mathematics, 29.10.2020 06:00


History, 29.10.2020 06:00

Mathematics, 29.10.2020 06:00

Mathematics, 29.10.2020 06:00

Mathematics, 29.10.2020 06:00

Advanced Placement (AP), 29.10.2020 06:00



Chemistry, 29.10.2020 06:00