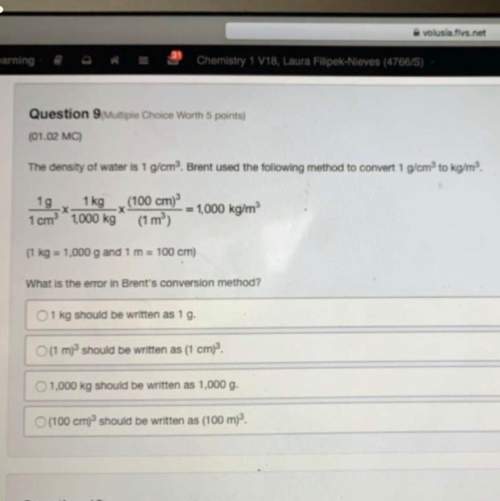
Chemistry, 23.06.2021 02:50 MathChic68
Which is true of the enthalpy of a reaction?
A. Its value depends on which reactions are added
B. Its value is the same as the enthalpy of each intermediate reaction
c. Its value is always a negative kJ/mol
D. Its value does not depend on which reactions are added

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 01:00
Which statement correctly describes potassium iodide, ki? there is a one-to-one ratio of potassium ions to iodide ions. potassium gains electrons and iodine loses electrons during the reaction. the lattice is held together by potassium anions and iodide cations.
Answers: 1

Chemistry, 22.06.2019 02:30
Asa choose the correct set of reaction coefficients to properly balance the following chemical equation according to the law of conservation of mass: __s8 + __o2 ==> __so2 1, 1, 8 1, 8, 1 1, 8, 8 8, 1, 1
Answers: 1

Chemistry, 22.06.2019 10:40
Ammonia and oxygen react to form nitrogen monoxide and water, like this: 4nh3 (g) + 5o2 (g) → 4no (g) + 6h2o (g) also, a chemist finds that at a certain temperature the equilibrium mixture of ammonia, oxygen, nitrogen monoxide, and water has the following composition: compound pressure at equilibrium nh3 65.1atm o2 31.3atm no 62.7atm h2o 65.8atm compound pressure at equilibrium nh3 65.3 atm o2 7.79 atm no 12.1 atm h2o 65.8 atm calculate the value of the equilibrium constant kp for this reaction. round your answer to 2 significant
Answers: 2

Chemistry, 22.06.2019 12:10
Building glycogen from glucose molecules is an example of
Answers: 3
You know the right answer?
Which is true of the enthalpy of a reaction?
A. Its value depends on which reactions are added
Questions


Biology, 12.10.2019 04:30



Mathematics, 12.10.2019 04:30

Mathematics, 12.10.2019 04:30


History, 12.10.2019 04:30

Mathematics, 12.10.2019 04:30

History, 12.10.2019 04:30

Mathematics, 12.10.2019 04:30





Chemistry, 12.10.2019 04:30


Health, 12.10.2019 04:30

Social Studies, 12.10.2019 04:30

World Languages, 12.10.2019 04:30