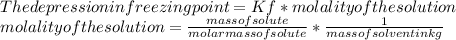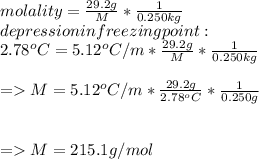Chemistry, 23.06.2021 02:50 jeffmacdonald1976
Suppose that you add 29.2 g of an unknown molecular compound to 0.250 kg of benzene, which has a K f of 5.12 oC/m. With the added solute, you find that there is a freezing point depression of 2.78 oC compared to pure benzene. What is the molar mass (in g/mol) of the unknown compound

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 04:40
*will mark you brainliest + 15 points ** why does the equilibrium of a system shift when the pressure is increased? a. to maximize the stress on the system b. to stop restoring equilibrium to the system c. to increase the total moles of gas in the system d. to decrease the total moles of gas in the system
Answers: 3

Chemistry, 22.06.2019 10:00
The tendency of water molecules to stick together is referred to as a) adhesion b) polarity c) cohesion d) transpiration e) evaporation
Answers: 1

Chemistry, 22.06.2019 11:00
The human eye contains a molecule called 11-cis-retinal that changes shape when struck with light of sufficient energy. the change in shape triggers a series of events that results in an electrical signal being sent to the brain that results in vision. the minimum energy required to change the conformation of 11-cis-retinal within the eye is about 164 kj/mol.
Answers: 2

You know the right answer?
Suppose that you add 29.2 g of an unknown molecular compound to 0.250 kg of benzene, which has a K f...
Questions


Mathematics, 11.05.2021 16:50





Mathematics, 11.05.2021 16:50

Biology, 11.05.2021 16:50



Mathematics, 11.05.2021 16:50



Mathematics, 11.05.2021 16:50


Physics, 11.05.2021 16:50

Social Studies, 11.05.2021 16:50

Health, 11.05.2021 16:50

Chemistry, 11.05.2021 16:50

Computers and Technology, 11.05.2021 16:50