A 3.81-gram sample of NaHCO3 was completely decomposed in an experiment.
2NaHCO3-Na2CO3 + H2CO3
In this experiment, carbon dioxide and water vapors combine to form H2CO3. After decomposition, the Na2CO3 had a mass of 2.86 grams.
A. Determine the mass of the H2CO3 produced.
B. Calculate the percentage yield of H2CO3 for the reaction. Show your work or describe the calculation process in detail.

Answers: 2
Another question on Chemistry

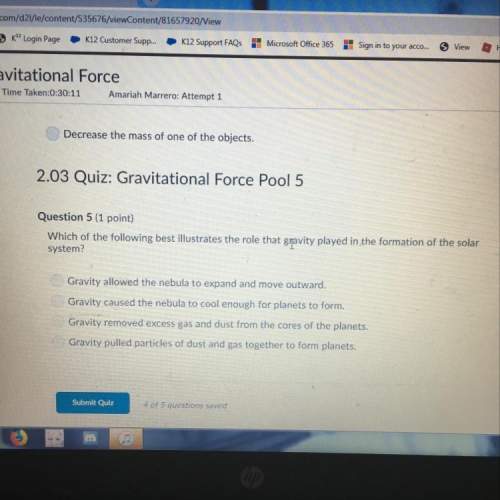
Chemistry, 22.06.2019 01:40
Which characteristic of water it form droplets? a. low specific heat b. nonpolar structure c. high surface tension d. ability to dissolve substances
Answers: 1

Chemistry, 22.06.2019 02:30
The is a particle with one unit of positive charge a. proton b. positron c. electron d. nucleus awnser quick it is a important science test!
Answers: 2

Chemistry, 22.06.2019 16:00
How does blood clotting prevent the entry of pathogens through cuts and wounds? answer asap,, this is due tomorrow. will mark as brainliest or whatever you call it : )
Answers: 2

Chemistry, 22.06.2019 22:30
Rank the four gases (air, exhaled air, gas produced from from decomposition of h2o2, gas from decomposition of nahco3) in order of increasing concentration of co2
Answers: 1
You know the right answer?
A 3.81-gram sample of NaHCO3 was completely decomposed in an experiment.
2NaHCO3-Na2CO3 + H2CO3
Questions

Social Studies, 06.10.2019 01:30







History, 06.10.2019 01:30



History, 06.10.2019 01:30

German, 06.10.2019 01:30

Mathematics, 06.10.2019 01:30

Spanish, 06.10.2019 01:30


Mathematics, 06.10.2019 01:30




History, 06.10.2019 01:30