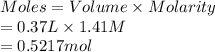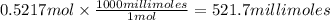Chemistry, 24.06.2021 15:50 brooklynpage5283
A chemist adds 370.0mL of a 1.41/molL potassium iodide KI solution to a reaction flask. Calculate the millimoles of potassium iodide the chemist has added to the flask. Be sure your answer has the correct number of significant digits.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 20:00
The picture represents the process that produces most of the energy used by living organisms on earth. which process is represented in the picture? a) the magnetic attraction between two hydrogen nuclei. b) the fusion of hydrogen nuclei to produce a helium nucleus in the core of the sun. c) the fission of hydrogen nuclei to produce a helium nucleus in the core of the sun. d) the chemical reaction between hydrogen nuclei to produce a helium nucleus in earth's atmosphere.
Answers: 3

Chemistry, 23.06.2019 00:40
To prevent the presence of air, noble gases are placed over highly reactive chemicals to act as inert "blanketing" gases. a chemical engineer places a mixture of noble gases consisting of 4.37 g of he, 13.36 g of ne, and 36.65 g of kr in a piston-cylinder assembly at stp. calculate the partial pressure in torr of kr.
Answers: 1


You know the right answer?
A chemist adds 370.0mL of a 1.41/molL potassium iodide KI solution to a reaction flask. Calculate th...
Questions

Mathematics, 19.08.2020 14:01

Mathematics, 19.08.2020 14:01


Mathematics, 19.08.2020 14:01

Mathematics, 19.08.2020 14:01


Chemistry, 19.08.2020 14:01

Mathematics, 19.08.2020 14:01


Mathematics, 19.08.2020 14:01

Biology, 19.08.2020 14:01

Mathematics, 19.08.2020 14:01

English, 19.08.2020 14:01


Mathematics, 19.08.2020 14:01

Mathematics, 19.08.2020 14:01

History, 19.08.2020 14:01


Mathematics, 19.08.2020 14:01