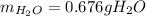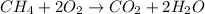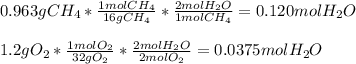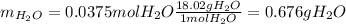Chemistry, 24.06.2021 17:20 celayaluis00
Gaseous methane CH4 will react with gaseous oxygen O2 to produce gaseous carbon dioxide CO2 and gaseous water H2O. Suppose 0.963 g of methane is mixed with 1.2 g of oxygen. Calculate the maximum mass of water that could be produced by the chemical reaction. Round your answer to 2 significant digits.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 13:00
16. why must the number of electrons lost equal the number of electrons gained in every redox reaction? use 3 – 4 sentences in your own words to address this question. 18. what type of radiation is emitted when chromium-51 decays into manganese-51? show the nuclear equation that leads you to this answer. 19. a radioactive nucleus alpha decays to yield a sodium-24 nucleus in 14.8 hours. what was the identity of the original nucleus? show the nuclear equation that leads you to this answer.
Answers: 2

Chemistry, 22.06.2019 13:50
How does the motion of particles in a gas change as the gas cools
Answers: 2

Chemistry, 22.06.2019 18:30
Which of the following words describe the reality that the universe looks the same from various perspective
Answers: 3

Chemistry, 22.06.2019 20:30
Consider the following unbalanced equation for the combustion of hexane: αc6h14(g)+βo2(g)→γco2(g)+δh2o(g) part a balance the equation. give your answer as an ordered set of numbers α, β, γ, use the least possible integers for the coefficients. α α , β, γ, δ = nothing request answer part b determine how many moles of o2 are required to react completely with 5.6 moles c6h14. express your answer using two significant figures. n n = nothing mol request answer provide feedback
Answers: 2
You know the right answer?
Gaseous methane CH4 will react with gaseous oxygen O2 to produce gaseous carbon dioxide CO2 and gase...
Questions




Mathematics, 02.10.2019 02:30

History, 02.10.2019 02:30





Social Studies, 02.10.2019 02:30

Mathematics, 02.10.2019 02:30


Mathematics, 02.10.2019 02:30





Mathematics, 02.10.2019 02:30