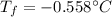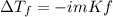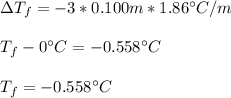Chemistry, 24.06.2021 20:20 daebreonnakelly
Calculate the freezing point of a 0.100 m aqueous solution of K2SO4, taking interionic attractions into consideration by using the van't Hoff factor (i for 0.100 m K2SO4

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:30
Summarize possible ways in which phases of matter could combine to form a solution.
Answers: 2


Chemistry, 22.06.2019 09:00
Suppose you have designed a new thermometer called the x thermometer. on the x scale the boiling point of water is 129 ? x and the freezing point of water is 13 ? x. part a at what temperature are the readings on the fahrenheit and x thermometers the same?
Answers: 1

You know the right answer?
Calculate the freezing point of a 0.100 m aqueous solution of K2SO4, taking interionic attractions i...
Questions



History, 10.06.2020 04:57

Mathematics, 10.06.2020 04:57

History, 10.06.2020 04:57




Health, 10.06.2020 04:57


Mathematics, 10.06.2020 04:57

Mathematics, 10.06.2020 04:57



English, 10.06.2020 04:57

Health, 10.06.2020 04:57