Chemistry, 25.06.2021 22:10 AmityHeart
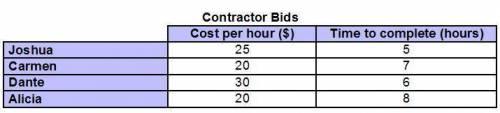
The chart shows the bids provided by four contractors to complete a job. Which contractor is the most cost-effective? Joshua Carmen Dante Alicia

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 03:20
Which type of substance ionizes partially and gives off hydrogen ions when dissolved in water? a. strong acid b. strong base c. weak acid d. weak base
Answers: 1

Chemistry, 22.06.2019 23:00
Consider the reaction: 2al(s) + fe2o3(s) → al2o3(s) + 2fe(s) the δhf for fe2o3(s) = -824.3 kj/mole. the δhf for al2o3(s) = -1675.7 kj/mole. finish the equation. δhrxn = [(1)( kj/mole) + (2)( kj/mole)] - [(1)( kj/mole) + (2) ( kj/mole)]
Answers: 1

Chemistry, 23.06.2019 00:00
The empirical formula of a compound is ch2o and its mass is 120 amu/molecule, what is its formula?
Answers: 1

Chemistry, 23.06.2019 00:20
Steam reforming of methane ( ch4) produces "synthesis gas," a mixture of carbon monoxide gas and hydrogen gas, which is the starting point for many important industrial chemical syntheses. an industrial chemist studying this reaction fills a 1.5 l flask with 3.5 atm of methane gas and 1.3 atm of water vapor at 43.0°c. he then raises the temperature, and when the mixture has come to equilibrium measures the partial pressure of carbon monoxide gas to be 1 .0 atm. calculate the pressure equilibrium constant for the steam reforming of methane at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 1
You know the right answer?
The chart shows the bids provided by four contractors to complete a job. Which contractor is the mos...
Questions



Mathematics, 24.05.2021 05:50

Mathematics, 24.05.2021 05:50



Mathematics, 24.05.2021 05:50





History, 24.05.2021 06:00

History, 24.05.2021 06:00


Mathematics, 24.05.2021 06:00


Mathematics, 24.05.2021 06:00


Mathematics, 24.05.2021 06:00