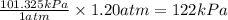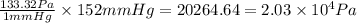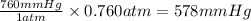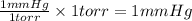Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 09:40
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3

Chemistry, 22.06.2019 15:00
Which substance is a steroid? cholesterol fatty acid monosaccharide trans fat
Answers: 1


Chemistry, 22.06.2019 22:50
At the current rate, a graph of carbon dioxide produced by fossil fuels over time would slope upward slope downward be horizontal be vertical
Answers: 3
You know the right answer?
Which of the following equations shows an incorrect relationship between pressures given in
terms o...
Questions

Mathematics, 06.10.2019 23:40

Arts, 06.10.2019 23:40


Mathematics, 06.10.2019 23:40


Mathematics, 06.10.2019 23:40


Mathematics, 06.10.2019 23:40

Mathematics, 06.10.2019 23:40


Social Studies, 06.10.2019 23:40

Advanced Placement (AP), 06.10.2019 23:40

Biology, 06.10.2019 23:40



Mathematics, 06.10.2019 23:40


Social Studies, 06.10.2019 23:40

Chemistry, 06.10.2019 23:40