
Chemistry, 28.06.2021 20:50 alexacarillo
Calculate [H3O+] and [OH−] for each of the following solutions at 25 ∘C given the pH.
pH= 11.22

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 03:10
Agas diffuses 1/7 times faster than hydrogen gas (h2). what is the molar mass of the gas? 100.10 g/mol 98.78 g/mol 86.68 g/mol 79.98 g/mol
Answers: 3

Chemistry, 22.06.2019 21:30
Isopropyl alcohol, (ch3)2choh, is a common solvent. determine the percent by mass of hydrogen in isopropyl alcohol. a) 6.71% h b) 13.4% h c) 25.0% h d) 53.3% h
Answers: 1

Chemistry, 22.06.2019 22:30
What is the value of the standard enthalpy of formation of an element in its most stable form?
Answers: 3

Chemistry, 23.06.2019 08:50
Reacting masses1 calcium carbonate breaks down on heating to produce calcium oxide and carbondioxide gas.caco3 + cao + co2a student heats 15 g of calcium carbonate strongly in a crucible.relative atomic masses (a): ca = 40, c = 12, o = 16.calculate the mass of calcium oxide produced by this reaction.(5 marks)
Answers: 3
You know the right answer?
Calculate [H3O+] and [OH−] for each of the following solutions at 25 ∘C given the pH.
pH= 11.2...
pH= 11.2...
Questions





Chemistry, 06.10.2020 01:01





World Languages, 06.10.2020 01:01

Spanish, 06.10.2020 01:01


Mathematics, 06.10.2020 01:01


Mathematics, 06.10.2020 01:01





Biology, 06.10.2020 01:01

![[H_{3}O^{+}]](/tpl/images/1385/4292/71b58.png) is
is  M and
M and ![[OH^{-}]](/tpl/images/1385/4292/e46dd.png) is
is 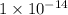 .
. ![pH = - log [H^{+}]\\11.22 = - log [H^{+}]\\conc. H^{+} = 6.025 \times 10^{-12}](/tpl/images/1385/4292/15de2.png) M
M![pOH = - log [OH^{-}]\\14 = - log [OH^{-}]\\conc. OH^{-} = 1 \times 10^{-14} M](/tpl/images/1385/4292/35504.png)



