Chemistry, 29.06.2021 02:10 fjjjjczar8890
Calculate the number of molecules of carbon (IV) oxide
produced when 10g of CaCO3 is treated with 100cm of
0.2mol dm HCl. The equation of the reaction is
CaCO3(s) + 2HCl(aq) → CaCl2(aq) + H2O(1) + CO2(g)
ICa=40 (=12 O=161

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 00:50
If a reactant was removed, did the new equilibrium system shift to make more reactants or more products?
Answers: 1

Chemistry, 22.06.2019 10:50
How many liters of oxygen gas, at standard temperature and pressure, will react with 35.8 grams of iron metal? 4 fe (s) + 3 o₂ (g) → 2 fe₂o₃ (s)
Answers: 2


Chemistry, 22.06.2019 22:40
Covalent bonds generally form when the bonded elements have a difference in electronegativity less than 1.5. subtract the electronegativities for the following pairs of elements and predict whether they form a covalent bond. electronegativity difference of c and c: ionic covalent electronegativity difference of mg and cl: ionic covalent
Answers: 1
You know the right answer?
Calculate the number of molecules of carbon (IV) oxide
produced when 10g of CaCO3 is treated with 1...
Questions

Mathematics, 06.05.2021 22:10

Mathematics, 06.05.2021 22:10






Mathematics, 06.05.2021 22:10


Geography, 06.05.2021 22:10



Mathematics, 06.05.2021 22:10



English, 06.05.2021 22:10


Biology, 06.05.2021 22:10

Mathematics, 06.05.2021 22:10

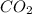 produced is 0.44 g
produced is 0.44 g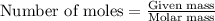 ......(1)
......(1)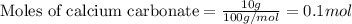
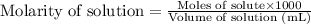 .....(2)
.....(2)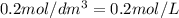 (Conversion factor:
(Conversion factor: 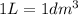
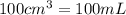 (Conversion factor:
(Conversion factor: 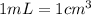
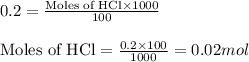
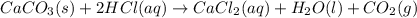
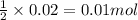 of calcium carbonate
of calcium carbonate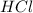 produces 1 mole of
produces 1 mole of