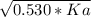Chemistry, 01.07.2021 21:30 jmcartwright00
An unknown weak acid with a concentration of 0.530 M has a pH of 5.600. What is the Ka of the weak acid

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3

Chemistry, 22.06.2019 11:30
Aperfume bottle is dropped in the corner of a room. the odor of the perfume can be detected on the other side of the room. which statement best describes this observation?
Answers: 2

Chemistry, 22.06.2019 12:20
Adeuteron, 21h, is the nucleus of a hydrogen isotope and consists of one proton and one neutron. the plasma of deuterons in a nuclear fusion reactor must be heated to about 3.02×108 k . what is the rms speed of the deuterons? express your answer using two significant figures.
Answers: 1

Chemistry, 22.06.2019 16:00
Answer asap : ( a. how does mucus prevent the entry of pathogens? b. describe two ways white blood cells protect us from pathogens.
Answers: 1
You know the right answer?
An unknown weak acid with a concentration of 0.530 M has a pH of 5.600. What is the Ka of the weak a...
Questions

Mathematics, 20.09.2020 14:01

Arts, 20.09.2020 14:01






Spanish, 20.09.2020 14:01






History, 20.09.2020 14:01

Computers and Technology, 20.09.2020 14:01

Mathematics, 20.09.2020 14:01

Biology, 20.09.2020 14:01

Mathematics, 20.09.2020 14:01


Computers and Technology, 20.09.2020 14:01

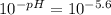 [H⁺] = 2.51x10⁻⁶ M
[H⁺] = 2.51x10⁻⁶ M