
Chemistry, 04.07.2021 22:10 KekePonds1021
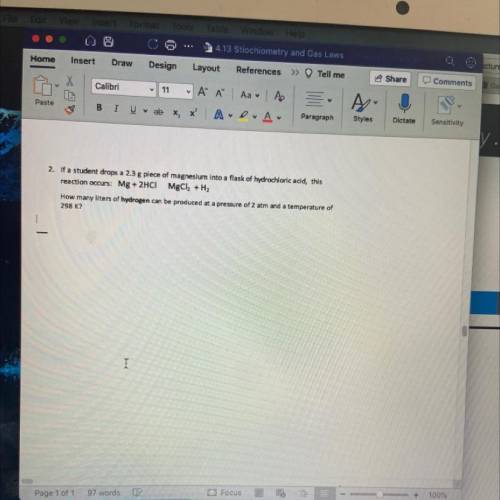
How many liters of hydrogen can be produced at a pressure of 2 atm and a temperature of 298 K

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:40
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2

Chemistry, 22.06.2019 13:00
The molality of calcium chloride (cacl2) in an aqueous solution is 2.46 m. what is mole fraction of the solute?
Answers: 3

Chemistry, 22.06.2019 13:00
12. calculate the hydroxide ion concentration of a solution with ph = 3.25. show all calculations leading to an answer
Answers: 3

Chemistry, 22.06.2019 19:30
Use the periodic table to find the molar mass of each element. molar mass h = g/mol molar mass s = g/mol molar mass o = g/mol
Answers: 3
You know the right answer?
How many liters of hydrogen can be produced at a pressure of 2 atm and a temperature of 298 K
...
...
Questions


Mathematics, 27.12.2020 09:30


Mathematics, 27.12.2020 09:30


Social Studies, 27.12.2020 09:30

Biology, 27.12.2020 09:30


Mathematics, 27.12.2020 09:30



Mathematics, 27.12.2020 09:30

Biology, 27.12.2020 09:30

History, 27.12.2020 09:30


Mathematics, 27.12.2020 09:30

Mathematics, 27.12.2020 09:40



Mathematics, 27.12.2020 09:40



