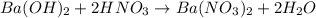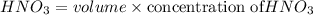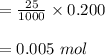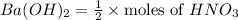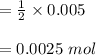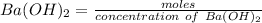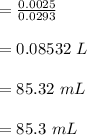Answer the following questions: (Questions about titration)
a. Why is it important to keep the NaOH solution covered at all times?
b. How will the molarity of NaOH solution be affected by its continued exposure to the atmosphere?
c. The pale pink color of the titration solution at the end point will fade to colorless after several minutes when exposed to the atmosphere. Account for this color change.
d. What volume (in mL) of 0.293 M Ba(OH)2 is required to neutralize 25.00 mL of 0.200M HNO3?

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 09:30
Which ocean zone has the most abundant primary producer and why a) the abyssopelagic zone ,du to the absence of light and cold water temperatureb) the bathypelagic zone, due to the absence of light and cold water temperaturec) the mesopelagic zone ,due to uts high light availability and warm water temperature d) the epipelagic zone,due to its high light availability and warm water temperature
Answers: 3

Chemistry, 22.06.2019 19:00
Which statement best describes what happens when molecular compounds melt
Answers: 1

Chemistry, 23.06.2019 05:30
For the reaction i2(g)+br2(g)←−→2ibr(g), kc=280 at 150 ∘c. suppose that 0.450 mol ibr in a 2.00-l flask is allowed to reach equilibrium at 150 ∘c. what is the equilibrium concentration of 2ibr, i2, br2
Answers: 1
You know the right answer?
Answer the following questions: (Questions about titration)
a. Why is it important to keep the NaOH...
Questions


Mathematics, 09.03.2021 21:00


Social Studies, 09.03.2021 21:00

History, 09.03.2021 21:00

Mathematics, 09.03.2021 21:00


Mathematics, 09.03.2021 21:00



Mathematics, 09.03.2021 21:00

English, 09.03.2021 21:00



Mathematics, 09.03.2021 21:00


Mathematics, 09.03.2021 21:00

Mathematics, 09.03.2021 21:00


Mathematics, 09.03.2021 21:00

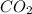 through dissolving in the solution and make carbonic acid
through dissolving in the solution and make carbonic acid 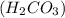 which reacts with the
which reacts with the 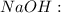
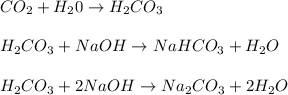
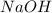 reacts with the dissolved
reacts with the dissolved 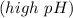 and colorless throughout the acidic solution
and colorless throughout the acidic solution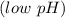 .
.
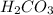 that gives the
that gives the 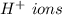
 lower pH.
lower pH.