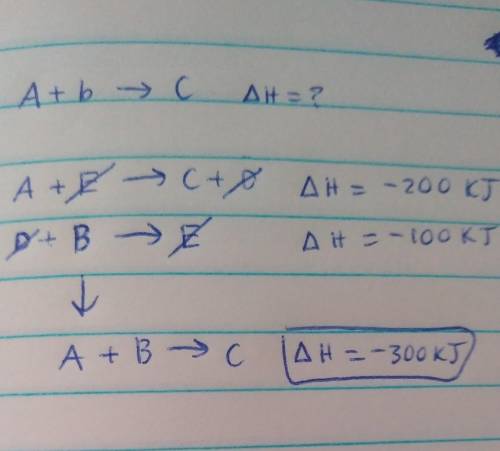Chemistry, 05.07.2021 23:20 anavallesdemiguel2
Determine the enthalpy for the reaction A+B --> C If we know the following:
A+E --> C+D; delta H = -200 kJ
D+B --> E; delta H = -100 kJ

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 15:00
Use examples from the article to explain one positive and one negative effect that chemistry has had on society
Answers: 2

Chemistry, 21.06.2019 17:40
What are the resulting coefficients when you balance the chemical equation for the combustion of ethane, c2h6? in this reaction, ethane is burned in the presence of oxygen (o2) to form carbon dioxide (co2) and water (h2o). (g)+(g)→(g)+(g)
Answers: 1

Chemistry, 21.06.2019 19:30
Complete the following reactions using word and balanced equations including states. dilute phosphoric acid is added with a calcium hydroxide solution.
Answers: 1

Chemistry, 22.06.2019 11:50
Which of the following statements about hybrid orbitals is or are true? choose all that apply. choose all that apply. under sp2 hybridization, the large lobes point to the vertices of an equilateral triangle. after an atom undergoes sp hybridization there is one unhybridized p orbital on the atom. the angle between the large lobes of sp3 hybrids is 109.5∘
Answers: 2
You know the right answer?
Determine the enthalpy for the reaction A+B --> C If we know the following:
A+E --> C+D; delt...
Questions




English, 08.01.2020 04:31

Mathematics, 08.01.2020 04:31

Mathematics, 08.01.2020 04:31






Mathematics, 08.01.2020 04:31


Mathematics, 08.01.2020 04:31


Computers and Technology, 08.01.2020 04:31



Mathematics, 08.01.2020 04:31

English, 08.01.2020 04:31