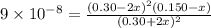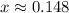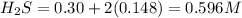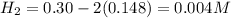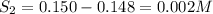For the equilibrium
2H2S(g) ⇋ 2H2(g) + S2(g) Kc = 9 .0X 10-8 at 700°C
the initial concentrati...

Chemistry, 06.07.2021 23:10 loganrose50
For the equilibrium
2H2S(g) ⇋ 2H2(g) + S2(g) Kc = 9 .0X 10-8 at 700°C
the initial concentrations of the three gases are 0.300 M H2S, 0.300 M H2, and 0. 1 50 M S2' Determine the equilibrium concentrations of the gases.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 12:10
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 13:30
The atomic number, or number, is the described as the number of in the nucleus of an chemical element.
Answers: 1

Chemistry, 22.06.2019 13:50
Read the chemical equation. 2c2h2 + 5o2 → 4co2 + 2h2o which of the following statements would be correct if one mole of c2h2 was used in this reaction? one mole of oxygen was used in this reaction. five moles of oxygen were used in this reaction. four moles of carbon dioxide were produced from this reaction. two moles of carbon dioxide were produced from this reaction.
Answers: 3

Chemistry, 23.06.2019 00:30
Maya wrote if you step to describe how carbon circulates between the atmosphere and living organisms
Answers: 1
You know the right answer?
Questions


English, 19.10.2021 14:00



Computers and Technology, 19.10.2021 14:00




SAT, 19.10.2021 14:00

Mathematics, 19.10.2021 14:00







Spanish, 19.10.2021 14:00

Mathematics, 19.10.2021 14:00


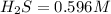
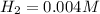
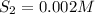
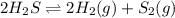
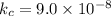
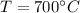
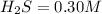
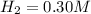
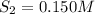
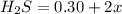
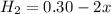
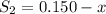
![K_c=\frac{product}{Reactant}=\frac{[H_2]^2[S_2]}{[H_2S]^2}](/tpl/images/1390/0282/9b893.png)