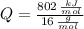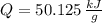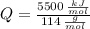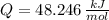Cuando se quema 1 mol de metano –o sea, 16 g–, se desprenden 802
kJ/mol.
○ Cuando se quema 1...

Cuando se quema 1 mol de metano –o sea, 16 g–, se desprenden 802
kJ/mol.
○ Cuando se quema 1 mol de octano –o sea, 114 g–, se desprenden 5500
kJ/mol.
Pareciera que el octano puede brindar más energía al quemarse, pero vamos a
hacer un análisis más cuidadoso. Comparemos la combustión de igual masa de
cada combustible.
● Calculen la cantidad de calor que se desprende cuando se quema un gramo
de cada combustible.
1 gr de metano aporta……………………………
1 gr de octano aporta………………………………

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 16:30
How does decreasing the gas volume affect the pressure of a gas?
Answers: 1


Chemistry, 22.06.2019 17:00
Which property of a rock remains unchanged by mechanical weathering? a. total surface area b. size and shape c. mineral composition d. sharpness
Answers: 1

Chemistry, 22.06.2019 18:10
The atom fluorine generally will become stable through the formation of an ionic chemical compound by accepting electron(s) from another atom. this process will fill its outer energy level of electrons.
Answers: 1
You know the right answer?
Questions



Computers and Technology, 21.08.2019 19:20




English, 21.08.2019 19:20

Mathematics, 21.08.2019 19:30




Mathematics, 21.08.2019 19:30


Social Studies, 21.08.2019 19:30

English, 21.08.2019 19:30


History, 21.08.2019 19:30

Mathematics, 21.08.2019 19:30


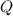 ), en kilojoules por mol, es igual a la cantidad de energía liberada por mol de compuesto (
), en kilojoules por mol, es igual a la cantidad de energía liberada por mol de compuesto (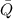 ), en kilojoules por mol, dividido por su masa molar (
), en kilojoules por mol, dividido por su masa molar (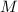 ), en gramos por mol:
), en gramos por mol: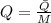 (1)
(1)