
Chemistry, 09.07.2021 03:20 yakshp3702
During a reaction, ΔH for reactants is −750 kJ/mol and ΔH for products is 920 kJ/mol. Which statement is correct about the reaction? (5 points)
Group of answer choices
It is endothermic because the energy required to break bonds in the reactants is less than the energy released when the products are formed.
It is endothermic because the energy required to break bonds in the reactants is greater than the energy released when the products are formed.
It is exothermic because the energy required to break bonds in the reactants is less than the energy released when the products are formed.
It is exothermic because the energy required to break bonds in the reactants is greater than the energy released when the products are formed.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 20:30
12. complete each of the following word equations for synthesis reactions. a. sodium + oxygen -> b. magnesium + fluorine -> 13. complete and balance the equations for the decomposition reactions. a. hgo -> [with the triangle heat symbol above the arrow] b. h2o(l) -> [with "electricity" written above the arrow]
Answers: 1

Chemistry, 22.06.2019 07:30
According to the vsepr theory what is the shape of a molecule that has a central atom valence three other items with no lone pairs of electrons
Answers: 1

Chemistry, 22.06.2019 10:30
Skills of homo sapiens were found an excavation. the skulls were preserved because the bodies were frozen. so, these fossils are (blank) fossils.the image shows the evolution of skulls beginning 2 to 3 million years ago. based on the image, modern human skulls(blank) ape skulls.
Answers: 1

You know the right answer?
During a reaction, ΔH for reactants is −750 kJ/mol and ΔH for products is 920 kJ/mol. Which statemen...
Questions



Mathematics, 17.09.2021 06:30

English, 17.09.2021 06:30

Mathematics, 17.09.2021 06:30


Mathematics, 17.09.2021 06:30



Mathematics, 17.09.2021 06:40

Mathematics, 17.09.2021 06:40

History, 17.09.2021 06:40


Chemistry, 17.09.2021 06:40


Mathematics, 17.09.2021 06:40




Mathematics, 17.09.2021 06:40

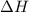 is always negative.
is always negative.

