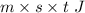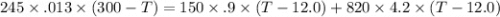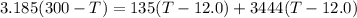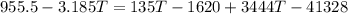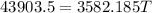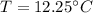Chemistry, 09.07.2021 19:40 zeesharpe05
What will be the equilibrium temperature when a 245 g block of lead at 300oC is placed in 150-g aluminum container containing 820 g of water at 12.0oC?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 16:00
Use the table to identify the phase and phase changes of the elements under the given conditions. write the name of the substance, phase, or phase change. when the temperature in a room increases from 25°c to 33°c, changes from a solid to a liquid. in a lab, methane and nitrogen are cooled from -170°c to -200°c. the methane freezes and the nitrogen . when gold is heated to 2,856°c it changes from a liquid to a .
Answers: 2

Chemistry, 21.06.2019 22:30
Check the correct box to describe the periodic trends in electronegativity. electronegativity across a period: decreases. increases. electronegativity down a group: decreases. increases.
Answers: 2

Chemistry, 22.06.2019 05:30
According to periodic trend, which of the following most likely has the highest ionization energy? kr be ni sc
Answers: 3

Chemistry, 22.06.2019 08:30
How would the number of moles (n) of o2 change if the atmospheric pressure doubled but all other variables stayed the same
Answers: 2
You know the right answer?
What will be the equilibrium temperature when a 245 g block of lead at 300oC is placed in 150-g alum...
Questions


English, 14.10.2020 01:01


Social Studies, 14.10.2020 01:01




Mathematics, 14.10.2020 01:01


Mathematics, 14.10.2020 01:01


Chemistry, 14.10.2020 01:01


Mathematics, 14.10.2020 01:01

Mathematics, 14.10.2020 01:01





Mathematics, 14.10.2020 01:01