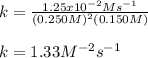NO2 (g) reacts with O2 4 NO2 (g) + O2(g) to produce N2 O5 (g) as shown by the equation below: (g) = 2 N2 O5 (g).
Experimentally the rate orders were determined and rate law written as shown below: Rate = k [NO2]2[O2 ].
Calculate the value of k if the initial concentration of NO2 was 0.250 M and initial
concentration of O2 (g) was 0.150 M. The initial rate was 1.25 x 10-2 M. s-1 .

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 18:30
In a sample of oxygen gas at room temperature, the average kinetic energy of all the balls stays constant. which postulate of kinetic molecular theory best explains how this is possible?
Answers: 2

Chemistry, 22.06.2019 07:30
11. phosphorus-32 is radioactive and has a half life of 14 days. how much of a 124 mg sample of phosphorus-32 is present after 56 days? a) 7.75 mg b) 15.5 mg c) 31.0 mg d) 62.0 mg
Answers: 3

Chemistry, 22.06.2019 18:30
How many moles of bromine are needed to produce 3.23 moles of potassium bromide
Answers: 1

Chemistry, 22.06.2019 22:30
Which of the following is not an assumption that scientists must make about the natural world? a. regularity b. causality c. predictability d. plausibility
Answers: 1
You know the right answer?
NO2 (g) reacts with O2 4 NO2 (g) + O2(g) to produce N2 O5 (g) as shown by the equation below: (g) =...
Questions


Social Studies, 18.08.2019 20:00

Mathematics, 18.08.2019 20:00



Mathematics, 18.08.2019 20:00


Physics, 18.08.2019 20:00

Mathematics, 18.08.2019 20:00


Mathematics, 18.08.2019 20:00




Mathematics, 18.08.2019 20:00


Chemistry, 18.08.2019 20:00

Mathematics, 18.08.2019 20:00

Mathematics, 18.08.2019 20:00

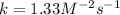
![k=\frac{r}{[NO_2]^2[O_2]}](/tpl/images/1392/3789/849e7.png)