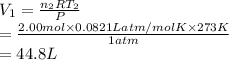Chemistry, 12.07.2021 05:40 mirzakasumovic8926
Consider an experimental run at 273 K where the initial number of moles (n1) is actually 1.00 mol, and the final number of moles (n2) is 2.00 mol. Use the simulation to find the volume (V1) of 1.00 mol of helium at 273 K, and calculate the final volume (V2). Express the volume to three significant figures, and include the appropriate units.

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 15:00
20 pts ‼️ an unmanned spacecraft travels to mars. mars has a lower strength of gravity than earth. where in the image is the spacecraft’s weight the greatest?
Answers: 1

Chemistry, 22.06.2019 23:10
Afusion reaction takes place between carbon and another element. neutrons are released, and a different element is formed. the different element is a) lighter than helium.b)heavier than helium.c)the same weight as helium.d)dependent on the element that reacted with carbon.
Answers: 3

Chemistry, 23.06.2019 02:30
What type of energy conversion occurs when you place your feet near the fire place and they become warm
Answers: 1
You know the right answer?
Consider an experimental run at 273 K where the initial number of moles (n1) is actually 1.00 mol, a...
Questions

Social Studies, 28.10.2019 12:31

Mathematics, 28.10.2019 12:31

Mathematics, 28.10.2019 12:31

Health, 28.10.2019 12:31


Mathematics, 28.10.2019 12:31

Mathematics, 28.10.2019 12:31

Mathematics, 28.10.2019 12:31

History, 28.10.2019 12:31

Mathematics, 28.10.2019 12:31

Computers and Technology, 28.10.2019 12:31

Mathematics, 28.10.2019 12:31



Biology, 28.10.2019 12:31

Mathematics, 28.10.2019 12:31

Chemistry, 28.10.2019 12:31



 of 1.00 mol of helium at 273 K is 22.4 L and the final volume
of 1.00 mol of helium at 273 K is 22.4 L and the final volume  is 44.8 L.
is 44.8 L. = 273 K,
= 273 K, 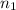 = 1.00 mol
= 1.00 mol = 273 K,
= 273 K,  = 2.00 mol
= 2.00 mol and
and 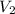 is calculated as follows.
is calculated as follows.