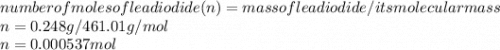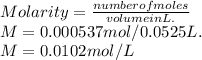Chemistry, 14.07.2021 01:20 omojay3103
If 52.5 mL of lead(II) nitrate solution reacts completely with excess sodium iodide solution to yield 0.248 g of precipitate, what is the molarity of lead(II) ion in the original solution

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 15:00
Which element in the third period would you expect to have the larger atomic radius, sodium (na) or sulfur (s)? a. sodium, because it has a higher effective nuclear charge attracting electrons in fewer energy levels. b. sodium, because it has fewer protons attracting electrons in the same energy levels. c. sulfur, because it has more protons attracting electrons in more energy levels. d. sulfur, because it has a higher effective nuclear charge attracting electrons in the same energy levels.
Answers: 2

Chemistry, 22.06.2019 01:00
What are the variables in gay-lussac’s law? pressure and volume pressure, temperature, and volume pressure and temperature volume, temperature, and moles of gas
Answers: 1

Chemistry, 22.06.2019 05:50
What happens when the temperature of a solution increases?
Answers: 2

Chemistry, 22.06.2019 19:30
To calculate percent by mass, use the equation below: calculate the percent by mass of each element. %n = % %h = % %o = %
Answers: 3
You know the right answer?
If 52.5 mL of lead(II) nitrate solution reacts completely with excess sodium iodide solution to yiel...
Questions


Mathematics, 14.10.2019 20:40



Mathematics, 14.10.2019 20:40


History, 14.10.2019 20:40



English, 14.10.2019 20:40



Spanish, 14.10.2019 20:40

Mathematics, 14.10.2019 20:40


Advanced Placement (AP), 14.10.2019 20:40




Computers and Technology, 14.10.2019 20:50