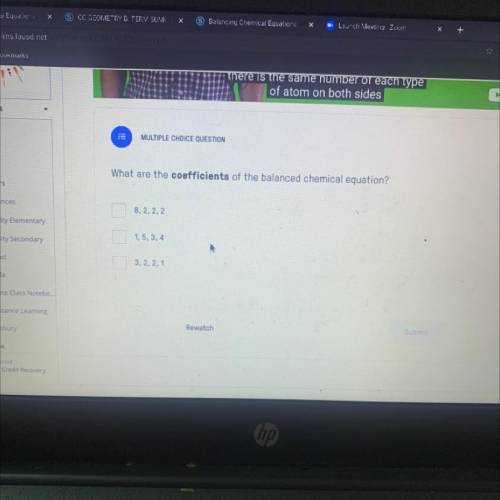
MULTIPLE CHOICE QUESTION
What are the coefficients of the balanced chemical equation?
8, 2, 2...

Chemistry, 14.07.2021 22:40 janeou17xn
MULTIPLE CHOICE QUESTION
What are the coefficients of the balanced chemical equation?
8, 2, 2, 2
1,5, 3, 4
3, 2, 2, 1
O...

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 21:00
Harvey kept a balloon with a volume of 348 milliliters at 25.0˚c inside a freezer for a night. when he took it out, its new volume was 322 milliliters, but its pressure was the same. if the final temperature of the balloon is the same as the freezer’s, what is the temperature of the freezer? the temperature of the freezer is kelvins.
Answers: 2

Chemistry, 22.06.2019 16:00
If 15 drops of ethanol from a medical dropper weight 0.60g, how many drops does it takes from a dropper to dispense 1.0ml of ethanol? the density of ethanol is 0.80g/ml
Answers: 1

Chemistry, 22.06.2019 18:00
Answer asap need to be answered by wednesday morning explain how a buffer works, using an ethanoic acid / sodium ethanoate system including how the system resists changes in ph upon addition of a small amount of base and upon addition of a small amount of acid respectively. include the following calculations in your i. calculate the ph of a solution made by mixing 25cm3 0.1m ch3cooh and 40cm3 0.1m ch3coo-na+. [ka = 1.74 x 10-5 m] ii. calculate the ph following the addition of a 10cm3 portion of 0.08 m naoh to 500cm3 of this buffer solution. iii. calculate the ph following the addition of a 10cm3 portion of 0.08 m hcl to 200cm3 of the original buffer solution.
Answers: 3

Chemistry, 22.06.2019 18:00
How many moles of oxygen gas are produced from the decomposition of six moles of potassium
Answers: 1
You know the right answer?
Questions


Spanish, 11.03.2020 02:54



Mathematics, 11.03.2020 02:54



Mathematics, 11.03.2020 02:54


History, 11.03.2020 02:54


Physics, 11.03.2020 02:54



Mathematics, 11.03.2020 02:54





Mathematics, 11.03.2020 02:54



