
Chemistry, 15.07.2021 21:50 avahrhey24
In aqueous solution the Ni2" ion forms a complex with four ammonia molecules. Write the formation constant expression for the equilibrium between the hydrated metal ion and the aqueous complex. Under that, write the balanced chemical equation for the first step in the formation of the complex K,=.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 21:40
During trial 2, what allowed you to determine that aluminum was the limiting reactant? check all that apply. all of the copper dissolved. all of the aluminum dissolved. the solution turned clear. the number of grams of copper(ii) chloride used in the reaction was greater than the number of grams of aluminum. the molar ratio of copper(ii) chloride to aluminum was greater than 3: 2, the equation’s molar ratio.
Answers: 2

Chemistry, 22.06.2019 16:30
Asample of freon gas has a volume of 2.23 liters, a pressure of 4.85 kpa, and a temperature of -1.36°c. calculate the volume at a pressure of 1.38 kpa and a temperature of 5.5°c. (show work)
Answers: 1


You know the right answer?
In aqueous solution the Ni2" ion forms a complex with four ammonia molecules. Write the formation co...
Questions


Computers and Technology, 09.12.2020 02:10

Mathematics, 09.12.2020 02:10



Mathematics, 09.12.2020 02:10






Mathematics, 09.12.2020 02:10


Mathematics, 09.12.2020 02:10

Computers and Technology, 09.12.2020 02:10


Mathematics, 09.12.2020 02:10

Mathematics, 09.12.2020 02:10

Health, 09.12.2020 02:10

![\frac{[Ni(H_2O)_3 (NH_3)]^{2+}}{[Ni(H_2O)_4]^{2+} [NH_3]}](/tpl/images/1394/7943/a809a.png) ".
".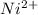 exist as
exist as ![[Ni(H_2O)_4]^{2+}](/tpl/images/1394/7943/49a05.png)
![[Ni(H_2O)_4]^{2+} + 4NH_3 \rightleftharpoons [Ni(NH_3)_4]^{2+} + H_2O](/tpl/images/1394/7943/ab9b4.png)
![K_f = \frac{[Ni(NH_3)_4]^{2+}}{[Ni(H_2O)_4^{2+}] [NH_3]^4}](/tpl/images/1394/7943/d666b.png)
![[H_2O]](/tpl/images/1394/7943/b5d70.png) as concentration of water will be const.
as concentration of water will be const.![[Ni(NH_3)_4]^{2+}](/tpl/images/1394/7943/dff73.png) proceeds via several steps,
proceeds via several steps,![[Ni(H_2O)_4]^{2+}+NH_3 \rightleftharpoons [Ni(H_2O)_3 (NH_3)]^{2+} + H_2O](/tpl/images/1394/7943/b2d29.png)
![K_1 = \frac{[Ni(H_2O)_3 (NH_3)]^{2+}}{[Ni(H_2O)_4]^{2+} [NH_3]}](/tpl/images/1394/7943/01675.png)


