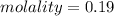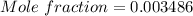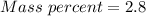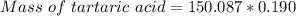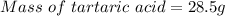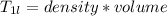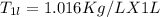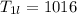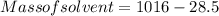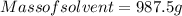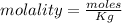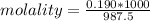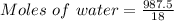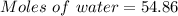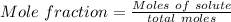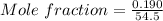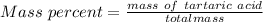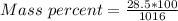Chemistry, 16.07.2021 01:00 911wgarcia
A beverage contains tartaric acid, H2C4H4O6, a substance obtained from grapes during wine making. If the beverage is 0.190 tartaric acid, what is the molal concentration? What is the mole fraction of tartaric acid and water? Calculate the mass percent of tartaric acid. The density of the solution is 1.016g/mL.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 13:00
What is common about these molecules? a.their atoms are held together by covalent bonds. b.they are all made up of the same two atoms. c.their atoms are held together by ionic bonds. d.they are all made up of oxygen atoms only.
Answers: 3


Chemistry, 22.06.2019 00:30
Water (2510 g ) is heated until it just begins to boil. if the water absorbs 5.09×105 j of heat in the process, what was the initial temperature of the water?
Answers: 3

You know the right answer?
A beverage contains tartaric acid, H2C4H4O6, a substance obtained from grapes during wine making. If...
Questions

Mathematics, 19.05.2021 16:40


Social Studies, 19.05.2021 16:40

Biology, 19.05.2021 16:40


Mathematics, 19.05.2021 16:40


Mathematics, 19.05.2021 16:40

Mathematics, 19.05.2021 16:40


Mathematics, 19.05.2021 16:40



Mathematics, 19.05.2021 16:40


Mathematics, 19.05.2021 16:40

Mathematics, 19.05.2021 16:40

Mathematics, 19.05.2021 16:40


Mathematics, 19.05.2021 16:40