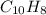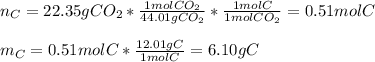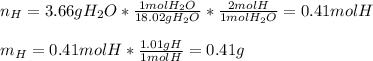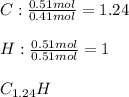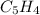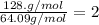Chemistry, 16.07.2021 02:30 ayoismeisjuam
6.50 g of a certain Compound x, known to be made of carbon, hydrogen and perhaps oxygen, and to have a molecular molar mass of 128. g/mol, is burned completely in excess oxygen, and the mass of the products carefully measured: product carbon dioxide water mass 22.35 g 3.66 g Use this information to find the molecular formula of x

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 20:00
Iam hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 1

Chemistry, 22.06.2019 22:30
Which of these statements best explains why space exploration should be encouraged? it prepares humans to live without oxygen. it dispel myths about objects in space. it prevents comets and asteroids from striking earth. it creates technology to absorb harmful radiations in space.
Answers: 1

Chemistry, 23.06.2019 08:00
At 35.0°c and 3.00 atm pressure, a gas has a volume of 1.40 l. what pressure does the gas have at 0.00°c and a volume of 0.950 l? which equation should you use? p2= p1v1t2/t1v2what is the pressure of the gas? 3.92 atm these are the answers
Answers: 1

Chemistry, 23.06.2019 16:30
There is a set up transformer that doubles the voltage. if the primary coil has a voltage of 10 v
Answers: 2
You know the right answer?
6.50 g of a certain Compound x, known to be made of carbon, hydrogen and perhaps oxygen, and to have...
Questions


Spanish, 17.09.2019 10:00

Physics, 17.09.2019 10:00

Biology, 17.09.2019 10:00




History, 17.09.2019 10:00



Biology, 17.09.2019 10:00



Computers and Technology, 17.09.2019 10:00

Biology, 17.09.2019 10:00


Social Studies, 17.09.2019 10:00



Social Studies, 17.09.2019 10:00