Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 02:30
When you perform this reaction, what could remain at the end of the reaction? check all that apply. excess reactant aqueous copper chloride excess reactant aluminum oxygen product solid copper carbon dioxide product aqueous aluminum chloride water
Answers: 2

Chemistry, 22.06.2019 04:50
Acompound contains c, h, and o atoms. when 1.130 g of the compound is burned in oxygen, 1.064 g co2 and 0.3631 g h2o are produced. what is the empirical formula of this compound?
Answers: 1

Chemistry, 22.06.2019 05:40
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2

Chemistry, 22.06.2019 15:10
Which statement describes the phase change that occurs when dry ice is placed in an open container at room temperature?
Answers: 1
You know the right answer?
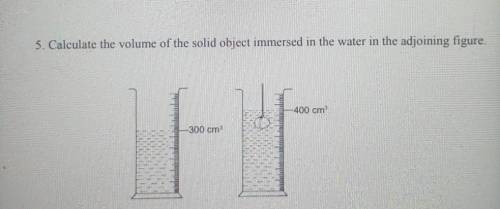
Calculate the volume of the object immersed in the water in the water in the adjoining figur.
Questions

Advanced Placement (AP), 26.02.2021 23:40





Mathematics, 26.02.2021 23:40


Mathematics, 26.02.2021 23:40

History, 26.02.2021 23:40

Biology, 26.02.2021 23:40

History, 26.02.2021 23:40

Physics, 26.02.2021 23:40


Mathematics, 26.02.2021 23:40



English, 26.02.2021 23:40


English, 26.02.2021 23:40