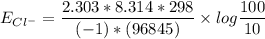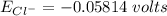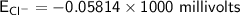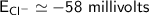The Nernst equation at 20oC is:
Eion= 58 millvolts/z. [log10 (ion)out/(ion)in]
Calculat...

Chemistry, 19.07.2021 17:10 shortty1111
The Nernst equation at 20oC is:
Eion= 58 millvolts/z. [log10 (ion)out/(ion)in]
Calculate the equilibrium potential for Cl- if the concentration of Cl- outside of the cell is 100 and the concentration inside of the cell is 10 mmol/liter.
a. 58 millivolts
b. +58 millivolts
c. -116 millivolts
d. 0

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 03:30
Melting and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 2

Chemistry, 22.06.2019 05:30
Liv sheldon given the balanced equation for an organic reaction: c2h2 + 2cl2 → c2h2cl4 this reaction is best classified as *
Answers: 1

Chemistry, 22.06.2019 06:00
An atom of sodium-23 (atomic number = 11) has a positive charge of +1. give this information, how many electrons does it have? how many proteins and neutrons does this atom have
Answers: 2

Chemistry, 22.06.2019 20:30
Some familiar products contain some of the same types of atoms. for instance, the chemical formula for baking soda is nahco 3. the chemical formula for liquid bleach is naclo, and the chemical formula for table salt is nacl. which choice best describes why these three products have some of the same types of atoms in common?
Answers: 1
You know the right answer?
Questions

Mathematics, 31.03.2021 21:00









English, 31.03.2021 21:00

Mathematics, 31.03.2021 21:00

English, 31.03.2021 21:00

Mathematics, 31.03.2021 21:00


Mathematics, 31.03.2021 21:00


Mathematics, 31.03.2021 21:00

Mathematics, 31.03.2021 21:00

Mathematics, 31.03.2021 21:00

![E_{ion} = 58 millivolts /z \Big[ log_{10} \Big( \dfrac{[ion]_{out}}{[ion]_{in}}\Big) \Big]}](/tpl/images/1396/2001/4bd00.png)
![E_{Cl^-} = \dfrac{2.303*R*T}{ZF} \times log \dfrac{[Cl^-]_{out}} {[Cl^-]_{in}}](/tpl/images/1396/2001/9486b.png)