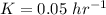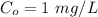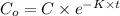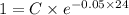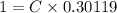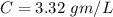Chemistry, 23.07.2021 02:20 Nathaliasmiles
A researcher is attempting to produce ethanol using an enzyme catalyzed batch reactor. The ethanol is produced from corn starch by first-order kinetics with a rate constant of 0.05 hr-1. Assuming the concentration of ethanol initially is 1 mg/L, what will be the concentration of ethanol (in mg/L) after 24 hours

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 15:30
Determine the empirical formula of a compound containing 40.6 grams of carbon, 5.1 grams of hydrogen, and 54.2 grams of oxygen. in an experiment, the molar mass of the compound was determined to be 118.084 g/mol. what is the molecular formula of the compound? for both questions, show your work or explain how you determined the formulas by giving specific values used in calculations.
Answers: 3

Chemistry, 21.06.2019 17:10
A+b→2c when the reaction begins, the researcher records that the rate of reaction is such that 1 mole of a is consumed per minute. after making changes to the reaction, the researcher notes that 2 moles of a are consumed per minute. what change could the researcher have made to effect this change?
Answers: 1

Chemistry, 22.06.2019 09:00
What is the percentage composition of carbon in the compound ch4
Answers: 1

Chemistry, 22.06.2019 14:30
The three types is stress that act on earths rocks are compression, tension, and
Answers: 1
You know the right answer?
A researcher is attempting to produce ethanol using an enzyme catalyzed batch reactor. The ethanol i...
Questions



English, 20.10.2020 21:01






Mathematics, 20.10.2020 21:01



Mathematics, 20.10.2020 21:01

Health, 20.10.2020 21:01


Mathematics, 20.10.2020 21:01


Mathematics, 20.10.2020 21:01


History, 20.10.2020 21:01

Mathematics, 20.10.2020 21:01