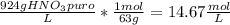Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 13:00
The number of neutrons is equal to the atomic number minus the atomic mass. a. true b. false
Answers: 2

Chemistry, 22.06.2019 13:30
1) which of the following is the best example of a physical change? a) sugar dissolving in tea b) firefly glowing 2) in the combustion of ethane, what is/are the reactants? c2h6 + o2 ==> co2 + h2o a) c2h6 and o2 b) co2 and c2h6
Answers: 2

Chemistry, 22.06.2019 15:30
How does a large body of water, such as the ocean, influence climate?
Answers: 1
You know the right answer?
Tenemos una disolución de HNO3 cuya pureza es del 65% y tiene una densidad de 1.32 g/mL. ¿Cuál es la...
Questions



World Languages, 04.10.2019 22:40

Chemistry, 04.10.2019 22:40


Mathematics, 04.10.2019 22:40


History, 04.10.2019 22:40


Social Studies, 04.10.2019 22:40

French, 04.10.2019 22:40


Geography, 04.10.2019 22:40


Mathematics, 04.10.2019 22:40

Mathematics, 04.10.2019 22:40

Business, 04.10.2019 22:40



Biology, 04.10.2019 22:40

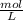 .
.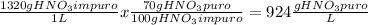
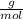 .
.