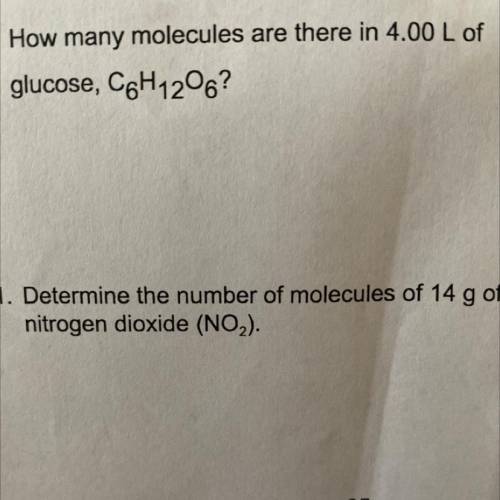How many molecules are there in 4.00 L of
glucose, C6H1206? Please show your work!!
...


Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:00
Ethanol (c2h5oh) is produced from the fermentation of sucrose in the presence of enzymes. c12h22o11(aq) + h2o(g) 4 c2h5oh(l) + 4 co2(g) determine the theoretical yield and the percent yields of ethanol if 680. g sucrose undergoes fermentation and 326.5 g ethanol is obtained. theoretical _ g _ percent %
Answers: 1

Chemistry, 22.06.2019 09:10
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

Chemistry, 22.06.2019 09:50
Achemist has dissolved a certain substance in water. the chemist knows that more of the substance could be dissolved into the water before it stops dissolving. therefore
Answers: 2

Chemistry, 22.06.2019 12:30
If anyone would be able to me out with these three questions it would be these are from the chem 2202 course.
Answers: 3
You know the right answer?
Questions




Mathematics, 08.10.2021 19:10



Social Studies, 08.10.2021 19:10

Biology, 08.10.2021 19:10




Biology, 08.10.2021 19:10


Geography, 08.10.2021 19:10


History, 08.10.2021 19:10

Mathematics, 08.10.2021 19:10



Mathematics, 08.10.2021 19:10