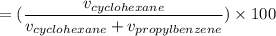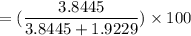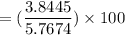Chemistry, 30.07.2021 01:30 janeou17xn
You perform a distillation to separate a mixture of propylbenzene and cyclohexane, and you obtain 2.9949 grams of cyclohexane (density -0.779 g/mL, MW - 84.16 g/mol) and 1.6575 grams of propylbenzene (density = 0.862 g/mL, MW = 120.2 g/mol). What is the volume percent composition of cyclohexane in the mixture?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 13:30
Mary is conducting an experiment on how pollution affects plant growth. how can she ensure that her data are reliable?
Answers: 3

Chemistry, 23.06.2019 02:20
Which of the following will cause an increase in the acceleration of an object? increase force decrease force increase mass decrease mass
Answers: 1

Chemistry, 23.06.2019 03:40
The following questions a24 - a26 relate to 100 ml of 0.0150 m solution of benzoic acid (c6h3cooh). ka(c6h3cooh) = 6.4 x 10^-5. what is the ph of the solution after the addition of 1 x 10^-3 moles of naoh? you may assume no volume change to the solution upon addition of the naoh.
Answers: 2

Chemistry, 23.06.2019 06:00
Complete the sentences to best explain the ranking.match the words below to the appropriate blanks in the sentences.a less polar bondhigher molar massion-dipole forcesstronger intermolecular forcesdipole-dipole forcesdispersion forceshydrogen bonding1. h2s and h2se exhibit the following intermolecular forces:.2. therefore, when comparing h2s and h2se the one with a has a higher boiling point .3. the strongest intermolecular force exhibited by h2o is . therefore, when comparing h2se and h2o the one with has a higher boiling point.
Answers: 1
You know the right answer?
You perform a distillation to separate a mixture of propylbenzene and cyclohexane, and you obtain 2....
Questions


Mathematics, 23.05.2021 17:10

World Languages, 23.05.2021 17:10


Computers and Technology, 23.05.2021 17:10



History, 23.05.2021 17:10

History, 23.05.2021 17:10

Mathematics, 23.05.2021 17:10


Mathematics, 23.05.2021 17:10


Mathematics, 23.05.2021 17:10



History, 23.05.2021 17:10


Mathematics, 23.05.2021 17:10