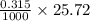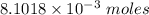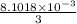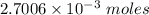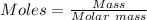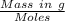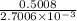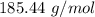Chemistry, 30.07.2021 19:50 elmoreroman11
0.5008 g of an unknown triprotic acid, H3A, is dissolved in 47.3 mL of water and then titrated with 0.315 M NaOH. It takes 25.72 mL of the NaOH solution to completely neutralize the acid. What is the molar mass of this acid

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 21:10
When the volume and number of particles of a gas are constant which of the following is also constant
Answers: 3

Chemistry, 22.06.2019 11:50
Which of the following statements about hybrid orbitals is or are true? choose all that apply. choose all that apply. under sp2 hybridization, the large lobes point to the vertices of an equilateral triangle. after an atom undergoes sp hybridization there is one unhybridized p orbital on the atom. the angle between the large lobes of sp3 hybrids is 109.5∘
Answers: 2

Chemistry, 22.06.2019 23:00
Which of your 24 wells had indications that a chemical reaction occurred? how were you able to tell that a chemical reaction occurred? which of your 24 wells had indications that a physical reaction occurred? how were you able to tell that a physical reaction occurred? report on both mixing and evaporation. make a general statement about whether your hypotheses were validated or rejected. must your hypotheses be correct for this to be a successful laboratory?
Answers: 3

Chemistry, 23.06.2019 01:30
How is the solubility of a carbon dioxide gas in water increase?
Answers: 1
You know the right answer?
0.5008 g of an unknown triprotic acid, H3A, is dissolved in 47.3 mL of water and then titrated with...
Questions




English, 16.04.2020 21:13






History, 16.04.2020 21:13




Mathematics, 16.04.2020 21:13


History, 16.04.2020 21:13

Biology, 16.04.2020 21:13

Mathematics, 16.04.2020 21:13