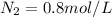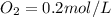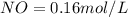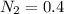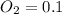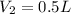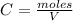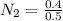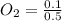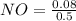Chemistry, 30.07.2021 22:50 Animallover100
A 1 L container originally holds 0.4 mol of N2, 0.1 mol of O2, and 0.08 mole of NO. If the volume of the container holding the equilibrium mixture of N2, O2, and NO is decreased to 0.5 L without changing the quantities of the gases present, how will their concentrations change

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 17:40
What are the resulting coefficients when you balance the chemical equation for the combustion of ethane, c2h6? in this reaction, ethane is burned in the presence of oxygen (o2) to form carbon dioxide (co2) and water (h2o). (g)+(g)→(g)+(g)
Answers: 1

Chemistry, 22.06.2019 01:50
7. what temperature is need to just dissolve 50 g of nh4cl in 75 g of water? '
Answers: 1

Chemistry, 22.06.2019 05:00
Which position represents spring in the southern hemisphere? a) b) c) d)
Answers: 2

You know the right answer?
A 1 L container originally holds 0.4 mol of N2, 0.1 mol of O2, and 0.08 mole of NO. If the volume of...
Questions















English, 13.11.2019 03:31

Computers and Technology, 13.11.2019 03:31