
It is often possible to change a hydrate into an anhydrous compound by heating it to drive off the water (dehydration). A 43.19 gram sample of a hydrate of MgBr2 was heated thoroughly in a porcelain crucible, until its weight remained constant. After heating, 27.21 grams of the anhydrous compound remained. What is the formula of the hydrate?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Complete the sentence. the lower the hydrogen ion concentration, the the ph. higher lower closer to 7 closer to 0
Answers: 2

Chemistry, 22.06.2019 00:30
This element exists in adundance in the sun.explain how you would go about capturing sunlight.would this captured sunlight contain any of the element?
Answers: 1

Chemistry, 22.06.2019 17:10
Increasing the substrate concentration in an enzymatic reaction could overcome which of the following? a) the need for a coenzymeb) allosteric inhibitionc) competitive inhibitiond) insufficient cofactors
Answers: 1

You know the right answer?
It is often possible to change a hydrate into an anhydrous compound by heating it to drive off the w...
Questions

History, 10.12.2019 03:31

Mathematics, 10.12.2019 03:31

Biology, 10.12.2019 03:31


Mathematics, 10.12.2019 03:31


Social Studies, 10.12.2019 03:31


History, 10.12.2019 03:31

English, 10.12.2019 03:31

Mathematics, 10.12.2019 03:31

Business, 10.12.2019 03:31

Mathematics, 10.12.2019 03:31



History, 10.12.2019 03:31




Biology, 10.12.2019 03:31

 .
. denote the number of
denote the number of 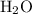 formula units for every
formula units for every  formula unit. The formula of the hydrate would be
formula unit. The formula of the hydrate would be  .
. :
: 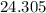 .
. :
: 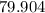 .
. :
:  .
. :
: 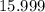 .
. .
. .
.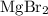 . There was
. There was 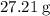 of this compound. Calculate the number of moles of formula units in that much of this compound:
of this compound. Calculate the number of moles of formula units in that much of this compound: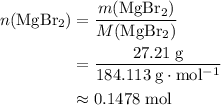 .
.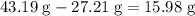 of water
of water 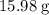 of
of 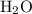 :
: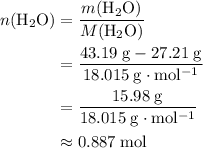 .
. of
of 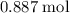 of
of 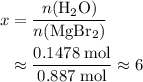 .
.

