In the following reaction, oxygen is the excess reactant.
SiCl4 + O2SiO2 + Cl2
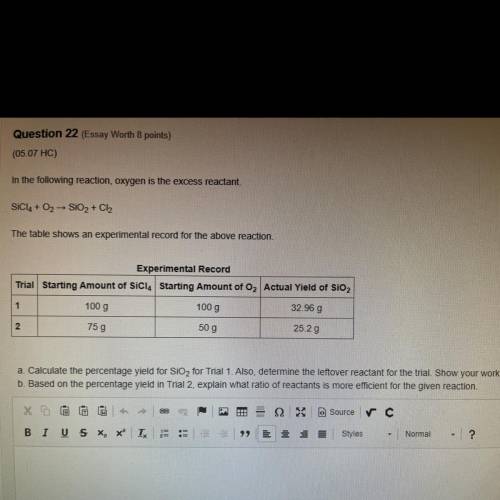
The table show...

In the following reaction, oxygen is the excess reactant.
SiCl4 + O2SiO2 + Cl2
The table shows an experimental record for the above reaction.
a. Calculate the percentage yield for SiO2 for Trial 1. Also, determine the leftover reactant for the trial. Show your work.
b. Based on the percentage yield in Trial 2, explain what ratio of reactants is more efficient for the given reaction.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 17:00
The atoms of a solid aluminum can are close together, vibrating in a rigid structure. if the can is warmed up on a hot plate, what happens to the atoms?
Answers: 3

Chemistry, 22.06.2019 23:00
Movement that is like a t a type of wave that transfers energy where the particles in the medium move in a circle motion while the energy travels left or right. a type of wave that transfers energy where the particles in the medium move perpendicular to the direction in which the energy is traveling. transfers energy from one location to another a type of wave that transfers energy where the particles in the medium move parallel to the direction in which the energy is traveling. movement that is back and forth, like an equal sign = 1. wave 2. parallel movement 3. perpendicular movement 4. transverse wave 5. longitudinal wave 6. surface wave
Answers: 1

Chemistry, 23.06.2019 02:00
Now look at the segment of the graph between the two data points marked with black squares. describe how the boiling point and melting point plots behave between these points. be as specific as possible.
Answers: 1
You know the right answer?
Questions


Mathematics, 23.10.2019 18:00


Health, 23.10.2019 18:00


Mathematics, 23.10.2019 18:00




Mathematics, 23.10.2019 18:00

Mathematics, 23.10.2019 18:00

History, 23.10.2019 18:00


Mathematics, 23.10.2019 18:00





Mathematics, 23.10.2019 18:00

Mathematics, 23.10.2019 18:00



