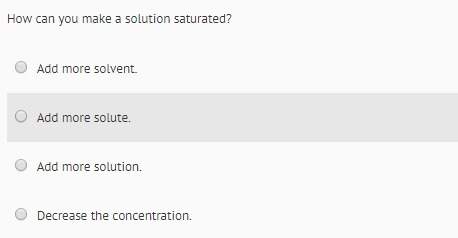
Chemistry, 21.08.2021 05:10 1Angel2Got3Brains
0.10 mol of NaOH is added to 0.30 mol of weak acid HX in a total volume of 1.00 L. The pH of the resultant solution is 8.65. What is Ka for HX

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 18:40
Determine the energy released per kilogram of fuel used. given mev per reaction, calculate energy in joules per kilogram of reactants. consider 1 mole of tritium plus 1 mole of deuterium to be a mole of “reactions” (total molar mass = 5 grams).
Answers: 1

Chemistry, 21.06.2019 19:10
Nuclear fusion is the source of energy for stars. besides hydrogen, which other element is most likely also common in stars?
Answers: 1

Chemistry, 21.06.2019 21:30
Aphysical reaction is a process in which one or more reactants change into one or more products with different properties. select the best answer from the choices provided t f
Answers: 1

Chemistry, 22.06.2019 11:00
Imagine that twenty i.u.’s of enzyme z were catalyzing the above reaction for one minute, under vmaxconditions, in a 3.00 ml assay volume. the assay is buffered with 20 mm phosphate buffer, ph 7.60. what will the ph be at the end of that one minute?
Answers: 2
You know the right answer?
0.10 mol of NaOH is added to 0.30 mol of weak acid HX in a total volume of 1.00 L. The pH of the res...
Questions

Social Studies, 07.08.2019 01:30






Social Studies, 07.08.2019 01:30




Mathematics, 07.08.2019 01:30





Engineering, 07.08.2019 01:30

English, 07.08.2019 01:30